Date: November 1, 2019 By:
A new approach that targets young immune cells may also work for creating vaccines against the flu, dengue, malaria and hepatitis C
October 31, 2019
LA JOLLA, CA
A collaborative team of scientists has made a successful proof-of-principle demonstration of an advanced HIV vaccine strategy—an approach that may also work in protecting people from an array of other deadly infectious diseases.
The team included scientists from Scripps Research, the Ragon Institute of Massachusetts General Hospital, MIT and Harvard, the La Jolla Institute for Immunology, and IAVI. Their research appears in Science.
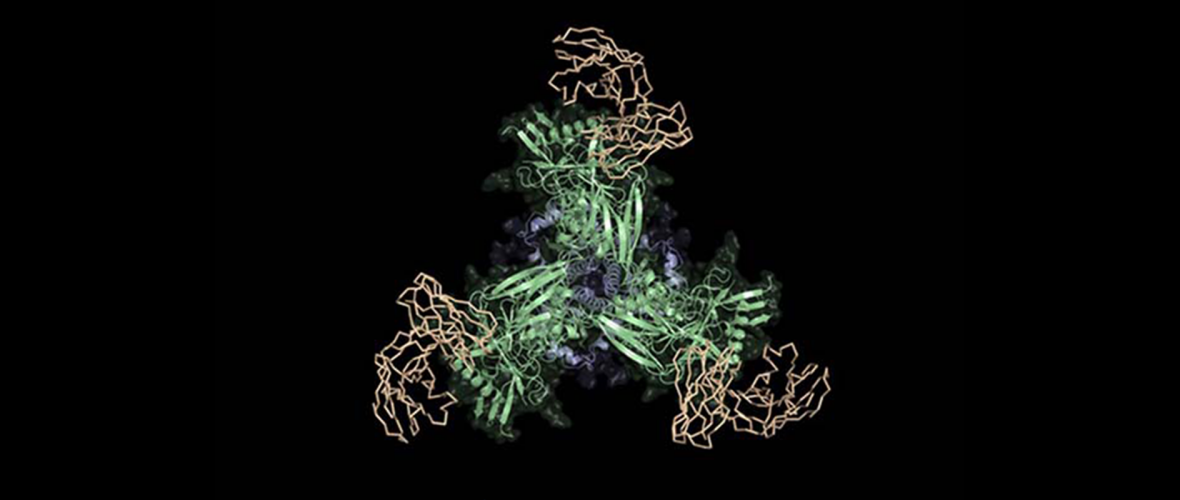
The new vaccine strategy centers on stimulating the immune system to produce broadly neutralizing antibodies (bnAbs) against HIV. These special antibodies are capable of neutralizing many different strains of the fast-mutating virus by binding to important yet difficult-to-access regions of the virus surface that don’t vary much from strain to strain.
A vaccine that elicits such antibodies could save many millions of lives and billions of dollars—and ultimately, may help eliminate HIV as a significant public health problem. Based on a concept called “germline targeting,” this novel strategy could potentially provide protection against the millions of different strains of the virus circulating globally. Achieving this goal has so far been elusive; no HIV vaccine candidate has ever been shown to induce a protective bnAb response in humans.
This study drew scientists from diverse backgrounds and areas of expertise: co-senior authors are Drs. William Schief, Professor in the Department of Immunology and Microbiology at Scripps Research, Facundo Batista, Chief Scientific Officer at the Ragon Institute, and Shane Crotty, Professor in the Vaccine Discovery Division at La Jolla Institute for Immunology.
“This is a very ambitious project that cannot be accomplished by one single lab. Instead, it requires a multi-disciplinary approach and can only succeed by virtue of collaboration,” says Facundo Batista, PhD.
“I believe that we need a germline-targeting strategy to develop an effective vaccine against HIV, and the same type of strategy may be helpful for making vaccines against many other difficult pathogens,” says William Schief, PhD, “Here, with a great collaborative effort among multiple labs, we’ve shown the feasibility of a general germline-targeting approach.”
The germline-targeting approach is meant to launch the production of a desired bnAb by stimulating the right antibody-producing cells. Antibodies are produced by immune cells called B cells, which start out in a “naïve” or “germline” state.
A large repertoire of these germline B cells circulates in the blood and other tissues. In a viral infection—or after immunization with a vaccine that mimics an infecting virus—some germline B cells will bind at least weakly to structures on the surface of the virus. That will stimulate the cells to begin a weeks-long maturation process, in which the antibodies continuously improve in their ability to bind to the surface, thereby neutralizing the virus.
The germline-targeting strategy for an HIV vaccine aims to stimulate the small number of germline B cells that are capable of maturing into cells that make bnAbs. Researchers suspect that other attempts to create an HIV vaccine that elicits bnAbs have failed because they haven’t stimulated a sufficient number of these “bnAb precursor” germline B cells.
To demonstrate the feasibility of their strategy, the authors started by choosing a known HIV bnAb called BG18. Informed by structural studies of BG18 bound to its target on HIV, they identified key features of the B-cell genes that encode this antibody’s HIV-gripping ability. Then, they searched a large database of antibody genes from circulating B cells in humans in order to find B cells that naturally share BG18’s key features. Choosing to target 28 of these potential “BG18-precursor” cells, they employed a sophisticated directed-evolution strategy to design a set of virus-mimicking proteins, called “immunogens,” that could stimulate such B cells in vaccine recipients.
“As the repertoire of circulating B cells differs from person to person, and even in the same person over time, we believe that it is important to target more than a few of these cells to have a reasonable chance of hitting one of them in any given vaccine recipient,” says Jon Steichen, PhD, a study co-first author and senior scientist in the Schief lab.
In the lab of Dr. Crotty, testing of blood samples from HIV-negative human donors confirmed that the team’s immunogens bound well to normal circulating B cells that have the desired BG18-like features. “Some of these B cells were exactly the type we were looking for,” says study co-first author Colin Havenar-Daughton, PhD, a senior scientist in the Crotty lab.
In the lab of Dr. Batista, experiments in mice engineered to carry BG18-precursor B cells showed that the immunogens could activate these B cells to induce potent antibody responses. “We generated a mouse model with rare BG18 precursors and high levels of competing B cells, mimicking physiological conditions encountered during human immunization. We found that our designed immunogens stimulated a strong and specific response from the bnAb precursors, and the elicited antibodies showed signs that they were maturing in the right direction”, says study co-first author Ying-Cing Lin, PhD, a postdoctoral fellow in the Batista laboratory.
The results of the study suggest that the team’s immunogens can work as an initial priming stage in a multi-stage vaccine aimed at eliciting BG18 and closely related bnAbs. The team now plans to develop immunogens for subsequent vaccine stages that will “shepherd” the antibody response to bnAbs. They envision an eventual HIV vaccine that combines several of these efforts to elicit multiple types of bnAbs against HIV.
Separate analyses performed in the study indicate that the same general germline-targeting method could be effective in designing vaccines to elicit bnAbs against influenza, dengue virus, Zika virus, hepatitis C and malaria.
The Scripps Research laboratories of Dennis Burton, PhD, Bryan Briney, PhD, and Ian Wilson, DPhil, also provided key assistance with genetic and atomic-structure analyses.
Support for the research was provided by the National Institute of Allergy and Infectious Diseases (UM1 Al100663, UM1 AI144462), the Ragon Institute, IAVI and the Bill and Melinda Gates Foundation.
Image courtesy of Scripps Research. Image description: Scientists at Scripps Research, the Ragon Institute, the La Jolla Institute for Immunology, and IAVI, designed a new vaccine candidate (green and blue) that binds to precursors of HIV broadly neutralizing antibodies (tan) and has promise as the first stage of an HIV vaccine.
About the Ragon Institute
The Ragon Institute of Mass General, MIT, and Harvard was established in 2009 with a gift from the Phillip T. and Susan M. Ragon Foundation, creating a collaborative scientific mission among these institutions to harness the immune system to combat and cure human diseases. A primary focus of the institute has been to contribute to the development of an effective AIDS vaccine. The Ragon Institute draws scientists and engineers from diverse backgrounds and areas of expertise across the Harvard and MIT communities and throughout the world to apply the full arsenal of scientific knowledge to understanding mechanisms of immune control and immune failure and to benefit patients. For more information about the Ragon Institute, visit www.ragoninstitute.org.
This article appears on the Ragon Institute website with the permission of the authors.
Ragon Institute Media contact: Corrie Martin, cmartin45@mgh.harvard.edu, 857-268-7074
Researchers at the Ragon Institute, including faculty member Hernandez Moura Silva, PhD, recently published a paper in Science Immunology with significant findings regarding resident tissue macrophages (RTMs), shedding light on their multifaceted roles in organ health.

After three years off due to the COVID-19 pandemic, the Ragon-MIT course HST.434 returned this January to provide 24 students a once in a lifetime learning experience

Ragon core member and MIT associate professor of chemical engineering Brandon DeKosky, PhD, was one of five MIT faculty members recently awarded $25 million to take on Cancer Grand Challenges.